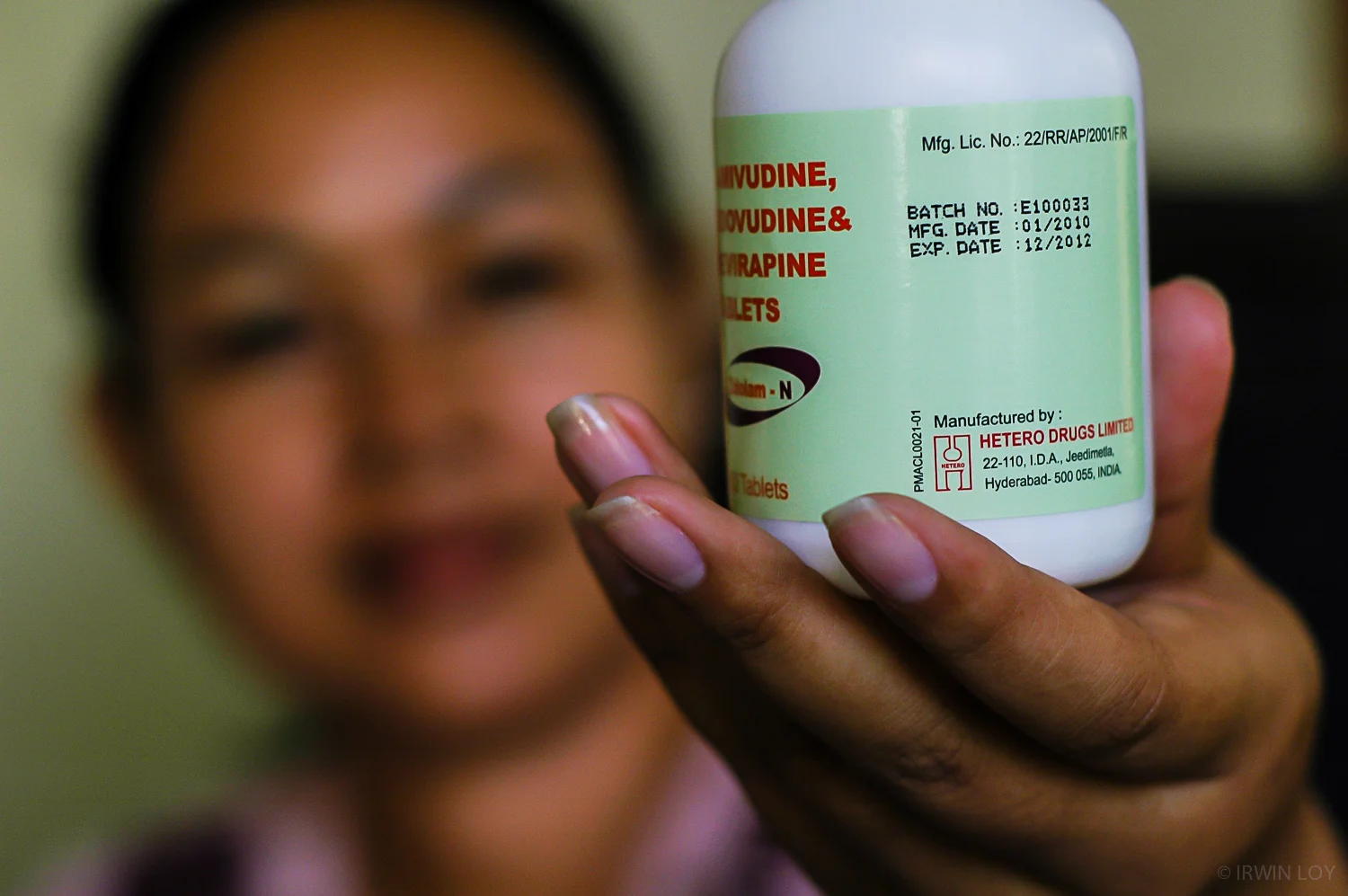
Every day, twice a day for the last seven years, Men Thol has swallowed a set of pills that gives him the strength to lead a normal life.
The 39-year-old tested positive for HIV in the mid 1990s. At first, he would rely on traditional medicines – usually ground up tree roots mixed with other vegetables and boiled in a broth.
“They never helped,” he said. “It was so terrible before. I always needed people to help me do things.”
He found his health became worse and worse. One year, he developed a prevalent skin rash over his body and he grew weaker and weaker. Finally in 2003, he was given a prescription for anti-retroviral (ARV) drugs, which are used to treat HIV. His condition stabilised and since then, he’s found a steady job and lives independently.
“My whole life is better than before,” he said. “I have energy and my health is fine. Now I can help my family, instead of the other way around.”
Thol is one of roughly 40,000 people living with HIV in Cambodia who are on life-saving ARV treatment.
However, advocates are warning that a pending trade deal between the European Union and India – a key supplier of ARVs to the developing world – could have a disastrous effect on those who need the drugs the most.
'We depend on the global community'
The EU and India are in the midst of negotiating a free trade agreement. But advocates for those living with HIV are worried a pact will include crippling new restrictions on intellectual property rights that, if enforced, will severely restrict access to the drugs by making it harder and more costly for Indian companies to produce generic medications.
“Restriction of generic drug production in India will have a devastating public health impact around the world.”
It’s no small issue in Cambodia, where more than 90 per cent of ARVs used in this South-east Asian nation are produced in India, according to Heng Phin, a programme manager with Cambodian People Living with HIV/AIDS Network, an advocacy group.
“This will have a big impact for people with HIV in Cambodia,” Phin said. “In Cambodia, we cannot produce ARV drugs. We depend on the global community and developed countries.”
Statistically speaking, Cambodia has taken significant strides in reducing its HIV prevalence rate and boosting the number of people on ARVs over the last 10 years.
The prevalence rate in the adult population, for example, has gone from a peak of 2 percent in 1998, to an estimated 0.7 percent this year, according to the country’s National Aids Authority.
And the number of people receiving crucial ARV treatment has exploded over the same period. There were only 71 patients being treated in 2001, according to the NAA. That number exceeded 40,000 by the middle of 2010, representing coverage of roughly 86 percent of the people who need access to such treatment.
Advocates say it’s no coincidence that over that time frame, the cost of buying ARVs has plummeted thanks to generic medicine produced in India.
“In 2000, it cost $10,000 per person to produce one year’s worth of ARV treatment. But now, that has been brought down to $80 per person,” Phin said.
“If you think that many people in Cambodia earn under $1 per day, you can see why we depend on generic medicine from India. If the cost is too high, then the government cannot buy it.”
Cambodia is merely representative of the concern over the current free trade negotiations.
Double standards
The group Médecins sans Frontières (MSF) says it depends on cheap generic medication to treat patients in 60 countries and that 80 percent of the HIV drugs it uses originate in India.
A report released last October by Oxfam International and Health Action International charges that the EU is “guilty of double standards”, by fighting to cut the price of medicine for European citizens, but in effect doing the opposite for people living with HIV in developing countries.
“The EU is pushing a range of [intellectual property] measures that would support the commercial interests of the pharmaceutical industry, while damaging the opportunity for innovation and access to medicines in developing countries,” the report argued.
However, the EU says any potential free trade pact will not be aimed at affecting India’s ability to export life-saving medication.
In a letter written this year and addressed to MSF, Karel De Gucht, the European Commissioner for trade, said the EC was “fully committed to ensuring that people in the world’s poorest countries can access affordable medicines.”
He said that key details over patent terms and data exclusivity were still to be negotiated, but that the EC was “ready to show the necessary flexibility” as discussions take place.
“I would like to be very clear that nothing in this agreement will prevent India from using compulsory licensing for manufacture and export of life-saving medicines to other developing countries in need,” De Gucht wrote.
But some aren’t as confident. In a statement released last week, Anand Grover, the United Nation’s special rapporteur on the right to health, warned that the potential trade deal “threatens” the production of life-saving medicine.
The free trade agreement in its current form – Grover referenced “leaked texts” of a draft FTA in his statement – will ensure that the production of generic medicines in India will be “severely hampered”, he contended.
“Millions in the developing world depend on India for generic medicines at affordable costs,” Grover said. “Restriction of generic drug production in India will have a devastating public health impact around the world and adversely affect the right to health of millions of patients.”
The EC and India officials say they hope to finalise an agreement by the spring of 2011.
In the meantime, however, people like Heng Phin worry what trickle down effects any pending agreement may have on him.
He is also HIV positive. And he remembers how getting on ARV treatment turned his life around a few years ago.
“I was very bad,” Phin recalled. “I was living in really bad conditions. My health was so poor. But after I got ARVs, then my health started getting better. I could work again.
“ARVs are the best. They are like gods that help people like me. The ARVs attack the virus so my health gets better. Now, I eat well. I can do anything.”
This article was first published by IPS.